Elucidating the mystery of phytoplasmas, the ultimate idler bacteria Basic research and fieldwork go hand-in-hand at the world’s oldest plant pathology laboratory

Always avoid being trapped by conventional wisdom. Professor Shigetou Namba smiles as he says, “Methods that everyone once thought heretical have, over the decades, become mainstream.” This spirit of inquiry has guided the Laboratory of Plant Pathology in the Department of Agricultural and Environmental Biology, Graduate School of Agricultural and Life Sciences, since it was founded in 1906 as the first such institution in the world. Today, that spirit underpins the laboratory’s research on tiny pathogenic microbes known as phytoplasmas, which cause disease in more than a thousand plant species and affect agricultural productivity worldwide.
The discovery of mycoplasma-like organisms, later referred to as phytoplasmas

Figure 1: Phytoplasma infection cycle
When a leafhopper inserts its piercing stylet into the vein of a plant infected with phytoplasmas and draws up the sap from the phloem, the phytoplasmas are transferred into the leafhopper’s body, where they multiply. Next, when the leafhopper feeds on sap from a healthy plant, phytoplasmas are introduced into the plant and the plant becomes infected.
© 2016 The University of Tokyo.
Once disease caused by phytoplasma is established, entire fields of crops might be wiped out. From cassava, the staple food of Southeast Asia, to grapes, the source of wine in Italy, and mulberry trees, the food for silk moths in China, these tiny micro-organisms are responsible for devastating damage to agricultural crops across the globe. “However, there are still no proven measures to address this problem,” says Namba.
Phytoplasmas are tiny bacteria about the size of a virus, one ten-millionth of a meter in length, but, unlike normal bacteria, they lack a cell wall. Transported by leafhoppers, close relatives of the cicada that are about two to three millimeters in length, phytoplasmas parasitize the phloem of leaves, stalks, and roots of plants, causing damage such as yellowing or death (figure 1). Because they obtain all their nutrients and energy from their host plants, these bacteria are also known as the “ultimate idlers.” First discovered by electron microscopy in 1967 by Emeritus Professor Yoji Doi, then a research student at the Laboratory of Plant Pathology, they were initially named mycoplasma-like organisms (MLO) because they resembled mycoplasma, a pathogenic bacterium of humans and other animals that also lacks a cell wall.
Molecular analysis resulted in a name change, from MLO to phytoplasma
Looking back on his choice of a research career, Namba recalls, “I chose the Laboratory of Plant Pathology because it was the most exciting place to be, with the discovery of MLOs and other activities there.” He later studied at Cornell University in the United States, where he acquired cutting-edge knowledge and expertise in rapidly-developing techniques in the life sciences, such as genetic analysis. Returning to Japan, he applied molecular biological techniques to the study of MLOs, taking research in this area to a new level.
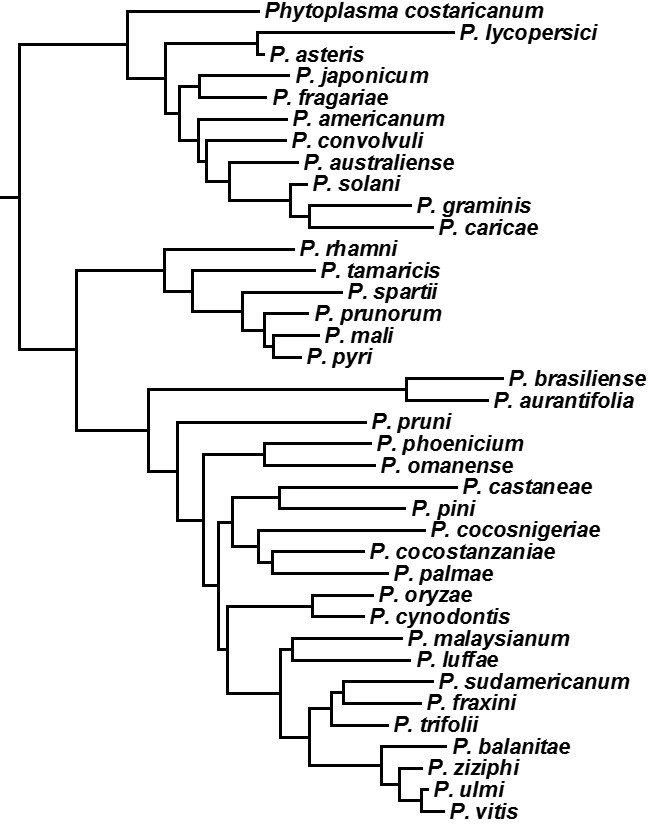
Figure 2: Classification of the Phytoplasma genus
Molecular phylogenetic classification of the Phytoplasma genus into 38 species, nine of which are found in Japan.
© 2016 Laboratory of Plant Pathology, Graduate School of Agricultural and Life Sciences, The University of Tokyo.
Research on MLOs had previously relied on examining diseased tissue with the naked eye or an electron microscope. It was nothing more than a conjecture that each particular form of disease was due to a unique MLO. A usable classification system was lacking. Beginning his studies of MLOs as an invited researcher with the Ministry of Agriculture, Forestry, and Fisheries, Namba used a laser to sever the stylets of leafhoppers that fed on phloem sap of rice plants infected with MLOs. He developed a fluorescent probe technique to detect the MLOs in the sap that oozed from the plants. Confident in his ability to succeed where others had failed, he purified the sap and devised a method to identify MLO genes and characterize them at the molecular level.
Namba next went on to identify nucleotide sequences of the MLO genes, in terms of their hereditary molecular differences. In 1993, he showed that the nucleotide sequences were different from those of mycoplasma. In 1995, he proposed the creation of the Phytoplasma genus as a new category of bacteria, and simplified the more than 1,000 phytoplasma species that had been defined according to the type of plants they infected to just 38 species (figure 2).
Metagenomic sequencing of the phytoplasma genome
A major obstacle stood in the way of continuing to sequence the entire genome. Because phytoplasmas were difficult to cultivate, routine techniques for their isolation, large-scale cultivation, and genome sequencing could not be utilized. Namba adopted a completely different approach. Rather than trying to purify a phytoplasma organism, he decided to extract the DNA from each infected plant, determine its nucleotide sequences, and then subtract the genome data for a healthy plant from that to derive the genomic sequences of the phytoplasma.
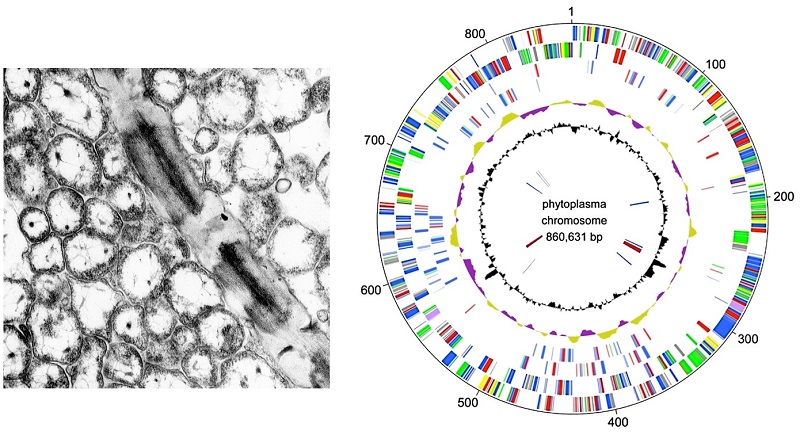
Figure 3: Electron micrograph (left) and genome map of phytoplasma (right)
(Left) Phytoplasmas (the roughly circular membrane-enclosed particles) are approximately one-ten millionth of a meter in size, only about one-tenth that of the giant virus known as pandoravirus. (Right) Its genome consists of approximately 870,000 base pairs, and has about one-third or one-quarter the number of genes of the pandoravirus.
© 2016 Laboratory of Plant Pathology, Graduate School of Agricultural and Life Sciences, The University of Tokyo.
Nowadays, this technique, known as metagenomic analysis, is used to sequence genomes of organisms such as gut flora and soil micro-organisms. At the time, the usual approach was to isolate and cultivate the target organism before sequencing the genome. “We decided on a strategy in which one genome would be subtracted from the other. When you stick to the conventional approach, a task can look impossible, but sometimes the only way to solve a problem is by tackling it head-on,” emphasized Namba.
In the midst of fierce competition from other research groups, Namba and his team led the world in 2004 by decoding the entire phytoplasma genome (figure 3). Next, his group elucidated the genes and their mechanisms of host specificity—how or why phytoplasmas colonize specific plants and insects—and identified the gene clusters and their functions that cause yellowing, death, and witches’ broom disease in plants. In short, by elucidating the pathogenic genes and their mechanisms, the group successfully answered many questions about phytoplasmas.
Plant research to benefit humanity
“I have always wanted to do research that benefits society. Rather than doing something to satisfy my wants, society’s needs are the topics of my research,” says Namba. The study of plant pathology was initially an academic discipline for solving problems that arise from this enormous field that affects society. The discovery of problems in the field, the solution of problems through basic research, and tackling new problems thereby discovered in the field; this has been the tradition of the laboratory that Namba continued, leading to multiple important discoveries.
Figure 4: Diagnostic kit for phytoplasma disease
Namba and his colleagues have developed a highly sensitive diagnostic kit that permits phytoplasma to be detected rapidly and easily that is now used for applications such as diagnosing witches’ broom disease of cassava, a major agricultural problem in Southeast Asia.
© 2016 Laboratory of Plant Pathology, Graduate School of Agricultural and Life Sciences, The University of Tokyo.
One of the results of this research was to develop a simple kit to diagnose the presence of a phytoplasma-borne disease. Namba and his colleagues developed and launched in 2011 a phytoplasma diagnostic kit that uses loop-mediated isothermal amplification, which is the highest-performing, ultra-sensitive gene amplification assay technique (figure 4). Using this kit, anyone can check whether a plant is infected with phytoplasma simply by cutting off a leaf, placing it in a tube containing special reagents, and heating to a boil. Next, adding 1 microliter of supernatant to the reaction tube and floating it on hot water allows one to determine, within 30 minutes, whether the plant is infected with phytoplasmas, with a sensitivity 100 times greater than the conventional polymerase chain reaction assays.
The kit could be used even in developing countries with inadequate experimental facilities. With support from the Japan International Cooperation Agency (JICA) and the Japan Science and Technology Agency (JST), Namba’s team has taken the technology to the local level. The kit is being used to assess coconut palm in Papua New Guinea. In 2015, the kit has been used as a diagnostic tool for cassava as a foodstuff and in bioethanol manufacturing in Southeast Asia. Training and professional development were also undertaken in tandem to establish the technology in each individual region.
Using pathogenicity to an advantage: enhancing our enjoyment of plants
Do plant pathogens such as phytoplasmas always have to play the role of the villain? Laboratory of Plant Pathology researcher Dr Misako Himeno remarked that “There is no doubt that phytoplasmas are pathogenic microorganisms that cause significant damage to agriculture, but if we can identify and successfully control their virulence factors, we can utilize them for good.”
Take the hydrangea, for example. Did you know that the hydrangea flower can turn into a leaf and become green in color? Because of their rarity, such hydrangeas are valuable and much sought after by enthusiasts (figure 5). However, this phenomenon is actually due to infection by phytoplasmas. The inspiration for Himeno’s laboratory research, the development of a flower into a leaf-like structure is called phyllody. In 2014, her colleagues discovered the protein secreted by phytoplasma that held the key to this process, naming it a phyllogen. As phyllogen degrades the protein that causes plant leaves to change into flowers, the flower is unable to develop normally, and reverts to a leaf structure.

Figure 5: A healthy hydrangea (left) and a hydrangea showing phyllody due to phytoplasma infection (right)
In research conducted by Himeno into the gene expression that occurs on the plant side when phyllody occurs, she found that the plant gene that changes leaves into flowers was only weakly expressed, and conjectured that phytoplasma causes phyllody by blocking the effect of this gene. This conjecture subsequently led to the discovery of the phyllogen protein.
© 2016 Laboratory of Plant Pathology, Graduate School of Agricultural and Life Sciences, The University of Tokyo.
While there are lovers of such phyllody-affected hydrangeas, there is the concern that the disease may spread into plants in agricultural areas, green spaces, or residential areas, because phytoplasmas are plant pathogens. Himeno counters that “If we can utilize the phyllogen protein to induce phyllody without causing disease, we may be able to readily grow ornamental green lilies and other flowers.”
For younger researchers such as Himeno, the traditions of the laboratory continue to unbroken. Initially interested in environmental issues, she entered the Faculty of Agriculture in the senior division. She became interested in the interactions between plants and micro-organisms, and this led to her involvement in phytoplasma research. Himeno continued her research while raising two children, but says, “As research is a team effort, we’ve been able to complete our experiments by helping each other.” For example, Himeno and her colleagues substitute for students who are unable to do lab work during the day because they are attending lectures. In turn, Himeno is supported by students in the lab when she cannot work in the evening, because she has to pick up her children from daycare.
Phytoplasmas cause agricultural damage across the globe, but the time might soon arrive when we can purchase vegetables and other plants that are made environmentally friendly, safe, and disease-resistant, thanks to the use of their pathogenic genes.
Interview/text: Katsue Nagakura
Translation: Tony Atkinson
Researchers

Professor Shigetou Namba

Dr. Misako Himeno
Links
Graduate School of Agricultural and Life Sciences
Laboratory of Plant Pathology, Graduate School of Agricultural and Life Sciences
Laboratory of Clinical Plant Science, Graduate School of Agricultural and Life Sciences (Japanese)
The University of Tokyo Plant Clinic (Japanese)
Japan Association of Clinical Plant Science (Japanese)






