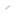A microRNA-mediated gene therapy for spinocerebellar ataxia Selective blockade of toxic protein translation

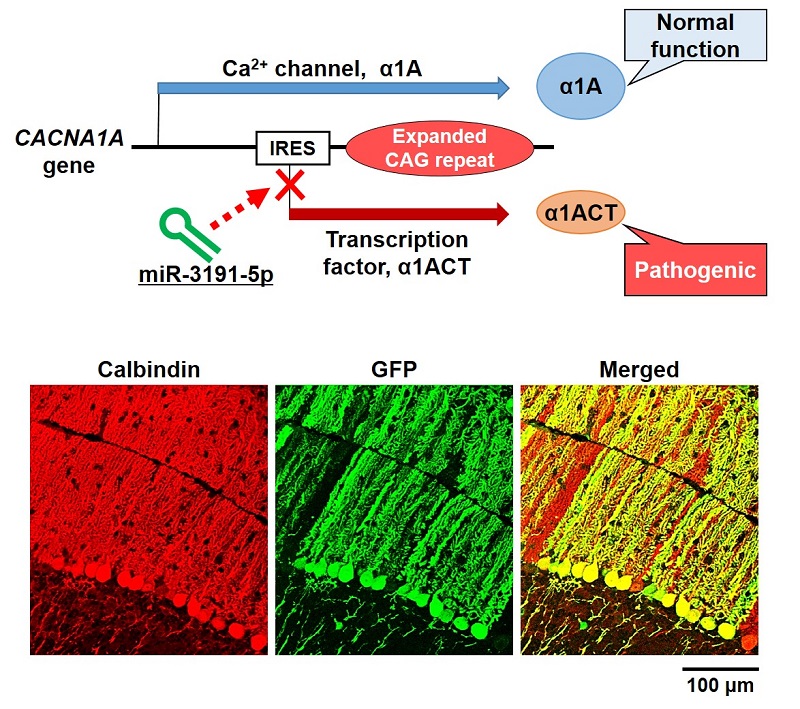
Selective suppression of α1ACT translation and delivery of miRNA into neuronal cells in the mouse cerebellum.
Upper panel: An miRNA (miR-3191-5p) selectively suppresses α1ACT translation while sparing calcium channel α1A expression.
Lower panel: Representative immunofluorescence images of the mouse cerebellum injected with adeno-associated virus vector expressing green fluorescence protein (GFP) and miR-3191-5p. Co-stained with calbindin-specific and GFP-specific antibodies. Calbindin-specific antibodies recognize Purkinje cells, large neurons in the cerebellum.
© 2016 Yu Miyazaki.
A research group from the University of Tokyo and the University of Chicago has developed a novel gene therapy for spinocerebellar ataxia type 6 (SCA6). SCA6 is a neurodegenerative disease affecting motor function in the patients. The researchers used a disease-specific microRNA (miRNA), a small RNA, to selectively inhibit the production of a disease-associated protein.
SCA is a neurodegenerative disease characterized by progressive impairment of motor function with neuronal cell loss in the cerebellum. Over 40 causative genes have been identified in SCAs so far; however, no disease modifying therapy is currently available. SCA6, the second-most common type of dominantly inherited SCAs in Japan, is caused by expanded repeat sequences in the CACNA1A gene. Although the CACNA1A gene has been thought to encode the calcium channel (αA1) necessary for neuronal cell activity, in 2013 a research group at the University of Chicago reported that, in addition to αA1, the CACNA1A gene also codes a second protein (α1ACT), a transcription factor, responsible for regulating gene expression, and that mutant α1ACT rather than αA1 causes neurodegeneration in SCA6.
Professor Shin-ichi Muramatsu from the Center for Gene and Cell Therapy, Institute of Medical Science, the University of Tokyo, and his collaborators have developed a novel therapeutic approach for SCA6 by selectively blocking α1ACT expression. They identified an miRNA that selectively inhibits the translation of α1ACT from CACNA1A messenger RNA. They used an improved adeno-associated virus (AAV) vector to efficiently deliver a disease-specific miRNA into the neuronal cells in SCA6 model mice. They also showed that an AAV vector-mediated miRNA delivery protected mice from the motor impairment and neuronal cell death in the cerebellum.
“A series of reports has recently suggested that miRNAs may hold therapeutic promise for treating cancer, metabolic diseases, and inflammation. We have previously demonstrated that an AAV vector-mediated miRNA delivery is effective in a mouse model of spinal and bulbar muscular atrophy, another type of neurodegenerative disease” says Muramatsu. He continues, “We are able to use our AAV vector systems to have a widespread transduction of genes throughout the brain and spinal cord in monkeys and pigs by an intrathecal administration. We would like to apply our strategy to the patients with SCA6 in clinics in the near future.”
This research was carried out in collaboration with Dr. Yu Miyazaki and Professor Christopher M. Gomez, from the Department of Neurology, the University of Chicago.
Paper
, "An miRNA-mediated therapy for SCA6 blocks IRES-driven translation of the CACNA1A second cistron", Science Translational Medicine Online Edition: 2016/07/14 (Japan time), doi: 10.1126/scitranslmed.aaf5660.
Article link (Publication)
Links
Center for Gene and Cell Therapy, Institute of Medical Science