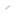New rechargeable battery utilizing redox between oxide and peroxide Innovative battery exceeds current lithium-ion batteries

The demand for rechargeable batteries is increasing as power sources not only for portable electronic appliances but also for large-scale applications such as electronic vehicles and stationary energy systems. Performance improvements are needed in terms of energy density, capacity, safety, and cost-effectiveness. Therefore, the development of innovative rechargeable batteries that are highly superior to current lithium ion battery systems is desired.
© 2014 Noritaka Mizuno.
Schematic representation of the new battery system operating on a redox reaction between oxide and peroxide. Redox reaction between lithium oxide and lithium peroxide takes place at the cathode, while redox reaction of metallic lithium takes place at the anode.
Professor Noritaka Mizuno at Department of Applied Chemistry, Graduate School of Engineering, the University of Tokyo and his research group collaborating with Nippon Shokubai Co., Ltd. have developed a new rechargeable battery system operating on a redox reaction between oxide and peroxide at the battery’s cathode. Its theoretical specific energy (the amount of energy that can be stored per unit of mass) is seven times as large as that of current lithium ion batteries based on the total weight of the cathode and anode active materials. The group demonstrated that peroxide species were formed in the charge process and consumed in the discharge process with cobalt-doped lithium oxide cathode material.
This new rechargeable battery system is capable of far larger specific energy and capacity than existing lithium-ion batteries. The battery system is expected to be a practical high-performance next-generation battery for electronic vehicles and stationary energy systems.
This research is supported by the Japan Society for the Promotion of Science (JSPS) through its “Funding Program for World-Leading Innovative R&D on Science and Technology (FIRST Program).”
Paper
Shin-ichi Okuoka, Yoshiyuki Ogasawara, Yosuke Suga, Mitsuhiro Hibino, Tetsuichi Kudo, Hironobu Ono, Koji Yonehara, Yasutaka Sumida, Yuki Yamada, Atsuo Yamada, Masaharu Oshima, Eita Tochigi, Naoya Shibata, Yuichi Ikuhara, Noritaka Mizunoa,
“A New Sealed Lithium-Peroxide Battery with a Co-Doped Li2O Cathode in a Superconcentrated Lithium Bis(fluorosulfonyl)amide Electrolyte”,
Scientific Reports Online Edition: 2014/7/14 (Japan time), doi: 10.1038/srep05684.
Article link
Links
Graduate School of Engineering
Department of Applied Chemistry, Graduate School of Engineering
Mizuno Laboratory, Department of Applied Chemistry, Graduate School of Engineering