Potential treatment strategy for Crohn's disease Shedding light on role of mesenteric fat in chronic intestinal inflammation

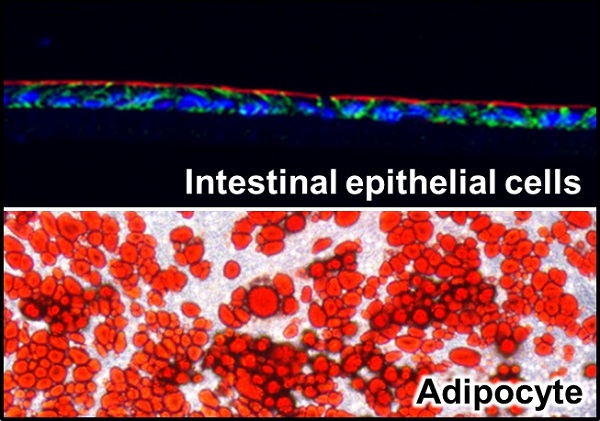
Co-culture of intestinal epithelial cells with adipocytes
The research group found that intestinal epithelial cells (top), prepared from intestinal tissue-like structures cultured outside the body, called organoids, and adipocytes (bottom), differentiated from mouse embryonic fibroblasts, induced a vicious inflammatory cycle when grown in the same medium—secretions from mature adipocytes trigger inflammation of the intestinal epithelial cells, and subsequently, secretions from inflamed epithelial cells induce inflammation of adipocytes. Inflammatory responses on both cells are believed to occur in a positive feedback cyclical manner.
© 2018 Yu Takahashi.
A group of University of Tokyo and Japan Tobacco Inc. researchers has revealed that the protective layer of cells, known as epithelial cells, lining the inner surface of the intestines, and fat cells, otherwise called adipocytes, trigger reciprocal inflammatory responses when these cells were cultured in the same medium. The current results suggest that adipocytes found in the intestinal membrane called the mesentery are directly involved in initiating and advancing intestinal inflammation, indicating that improvement of adipocyte characteristics could lead to new treatment strategies for Crohn's disease, a type of chronic inflammatory bowel disease.
Researchers have recognized since the 1930s that increased mesenteric fat tissues are frequently observed adjacent to breaks in the mucous membrane. Scientists have also reported that inflammation occurs in the increased mesenteric fat tissues. However, there has been little direct evidence so far linking mesenteric fat to intestinal inflammation, and the way in which increased, inflamed mesenteric fat tissues contributes to the inflammation of intestinal epithelial cells has very seldom been elucidated.
In the current study, the research group led by Visiting Associate Professor Shintaro Sato and Professor Hiroshi Kiyono at the Institute of Medical Science at the University of Tokyo and researcher Yu Takahashi at Japan Tobacco Inc. prepared their own intestinal epithelial cells, consisting of various cells found in the intestines. When these epithelial cells were co-cultured with differentiated adipocytes, expression of pro-inflammatory genes increased, while anti-inflammatory gene expression decreased. Moreover, the group found that the inflammatory responses were mediated by proteins regulating the transcription of genes, called transcription factors, such as the NF-κB and STAT3 signaling pathways, by using specific inhibitors.
From their analysis, the researchers found that the inflammatory signals from adipocytes could be transmitted to intestinal epithelial cells directly. Abnormality of mesenteric fat tissues is thought to occur from the onset of Crohn's disease; therefore, the present findings suggest that inflammatory responses on adipocytes could contribute to the onset and/or exacerbation of Crohn's disease. In addition, the researchers indicate that modifying a certain property of mesenteric fat tissues has the potential of leading to new therapies for Crohn's disease.
"Given that suppression of inflammatory responses in mesenteric adipocytes can ameliorate intestinal inflammation will lead to the development of novel drugs and vaccines," says Kiyono. He continues, "Like this study, future investigations to elucidate the cause of a certain disease or to develop treatment strategies will become more and more focused on the intercellular and the relationships among cells. Our study is meaningful in terms of pioneering a new research field."
Paper
, "Reciprocal inflammatory signaling between intestinal epithelial cells and adipocytes in the absence of immune cells", EBioMedicine Online Edition: 2017/08/02 (Japan time), doi: 10.1016/j.ebiom.2017.07.027.
Article link (Publication)
Links
Department of Microbiology and Immunology, Institute of Medical Science