Roles of common subunits within distinct multisubunit complexes Method of determining distinct functions

The same protein is often a subunit of more than one multisubunit protein complex, each of which has a distinct function within cells. A mutation in such a protein would cause multiple cellular defects; therefore, it is difficult to distinguish between the function of a protein in one complex from its functions in other complexes. Associate Professor Masami Horikoshi at the University of Tokyo Institute of Molecular and Cellular Biosciences, Professor Masayuki Seki at Tohoku Pharmaceutical University and their colleagues have developed a unique strategy to overcome this problem, which will help to analyze a variety of biological processes by revealing the specific roles played by such proteins within multisubunit protein complexes.
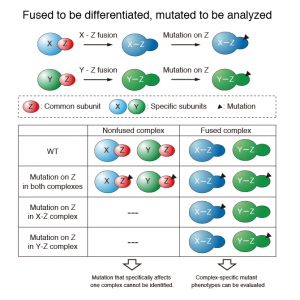
Currently, there is no method to distinguish between the roles of a subunit in one multisubunit protein complex from its roles in other complexes in vivo. This is because a mutation in a common subunit will affect all complexes containing that subunit. The authors describe a unique method (FALC: Functional Analysis of Linker-mediated Complex) to discriminate between the functions of a common subunit in different multisubunit protein complexes. In this method, a common subunit in a multisubunit protein complex is genetically fused to a subunit that is specific to that complex and point mutated. The resulting phenotype(s) identify the specific function(s) of the subunit in that complex only. Histone H2B is a common subunit in nucleosomes containing H2A/H2B or Htz1/H2B dimers. The H2B was fused to H2A or Htz1 and point mutated. Their strategy revealed that H2B has common and distinct functions in different nucleosomes.
This method could be used to study common subunits in other multisubunit protein complexes.
Press release (Japanese)
Paper
Yu Nakabayashi, Satoshi Kawashima, Takemi Enomoto, Masayuki Seki and Masami Horikoshi,
“Roles of common subunits within distinct multisubunit complexes”,
Proceedings of the National Academy of Sciences 111 (2014) : 699-704, doi: 10.1073/pnas.1316433111.
Article link
Links
Institute of Molecular and Cellular Biosciences
Laboratory of Developmental Biology, Institute of Molecular and Cellular Bioscience (Japanese)