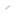Simultaneous translation of two distinct peptides from single mRNA template Engineered translation system works independently from natural system

Ribosomes in which proteins are synthesized and tRNAs (transfer RNAs) that transfer amino acids to them play the central role in translation, the process by which peptides are synthesized from an mRNA (messenger RNA) template according to a genetic code. Three bases each from the rRNA (ribosomal RNA) that comprises the ribosome and from tRNA bind together to create complementary base pairs (guanine (G) and cytosine (C) in the image). It has been suggested that these three base pairs are essential for peptide synthesis as amino acids bind together which are transfered by tRNA inside the ribosome.
© 2014 Hiroaki Suga.
Both wild-type (WT) and engineered ribosome-tRNA pairs were mixed with a single type of mRNA. The WT ribosome-tRNA pair produced a peptide containing natural amino acids following WT genetic code, and the engineered pair produced a peptide containing non-natural amino acids following an artificial genetic code.
Professor Hiroaki Suga’s group in the Department of Chemistry in the Graduate School of Science constructed a range of ribosome-tRNA pairs with complementary mutations in order to preserve the base pairs between the two RNA types, and analyzed their translation activities. Consequently, a new translation system working independently to the wild-type system according to an artificially designed genetic code was successfully established by using one of the mutant ribosome-tRNA pairs.
In addition, it became possible to express two different peptides from the same gene by charging different amino acids to the mutated tRNAs than to the wild type tRNAs. In effect, the research group developed a modified translation system that works from an artificially designed genetic code.
In this research, the research group revealed the importance of the base pair interactions between rRNA and tRNAs in the peptidyl-transfer center, and developed a novel method for genetic code reprogramming. This method can be applied to the synthesis of non-standard peptides containing non-proteinogenic amino acids, which has potential applications in drug development.
Press release (Japanese)
Paper
, "An orthogonal ribosome-tRNAs pair via engineering of the peptidyl transferase center", Nature Chemical Biology Online Edition: 2014/6/9 (Japan time), doi:10.1038/NCHEMBIO.1549.
Article link (Publication, UTokyo Repository)
Links
Department of Chemistry, Graduate School of Science
Laboratory of Bioorganic Chemistry, Department of Chemistry, Graduate School of Science