Toward the development of effective therapeutics against Alzheimer’s disease Identification of molecular mechanism of γ-secretase modulators

Alzheimer’s disease (AD) is the most common dementing disorder and is one of biggest problems facing aging societies. It is thought to be caused by a deposition of Amyloid-β peptide (Aβ) in the brain as senile plaques. In particular, the aggregation-prone species Aβ42 is thought to be the molecular culprit for AD. γ-Secretase is an enzyme responsible for Aβ generation. Thus, γ-secretase modulators, which specifically inhibit Aβ42 generation, have been developed as therapeutics against AD without side-effects. However, molecular mechanism whereby γ-secretase modulators regulate enzymatic activity remained unknown.
© 2014 Taisuke Tomita.
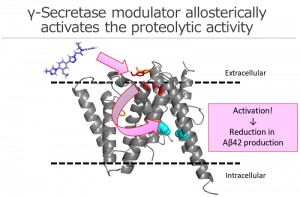
Phenylimidazole-type γ-secretase modulator targets to the extracellular side of presenilin, a catalytic subunit, and allosterically induces a conformational changes at the intramembrane catalytic site to reduce Aβ42 production from neurons.
Researchers lead by Professor Taisuke Tomita, Ph.D., and graduate student Koji Takeo, Ph.D., at the Graduate School of Pharmaceutical Sciences, the University of Tokyo, identified that phenylimidazole-type γ-secretase modulators activate the proteolytic activity of γ-secretase. Moreover, they found that the compound targets to the extracellular region of presenilin, an enzymatic subunit of γ-secretase, to allosterically (indirectly bring about a change by binding to a protein) induce conformational changes at the catalytic site.
This is the first report to identify the molecular mechanism and binding site of the phenylimidazole-type γ-secretase modulator. These findings would lead to the rational design of γ-secretase modulators with better potency based on the structure and function of the molecule, and its interaction with the target, and the discovery of effective therapeutics against AD.
Paper
Takeo K, Tanimura S, Shinoda T, Osawa S, Zahariev IK, Takegami N, Ishizuka-Katsura Y, Shinya N, Takagi-Niidome S, Tominaga A, et al.,
“Allosteric regulation of γ-secretase activity by a phenylimidazole-type γ-secretase modulator”,
Proceedings of the National Academy of Sciences Online Edition: 2014/7/9 (Japan time), doi: 10.1073/pnas.1402171111.
Article link
Links
Graduate School of Pharmaceutical Sciences
Department of Neuropathology and Neuroscience, Graduate School of Pharmaceutical Sciences