Cycle of changes inside the cell Autophagy research holds great promise for future advances in medicine

A research field taking off in the last decade
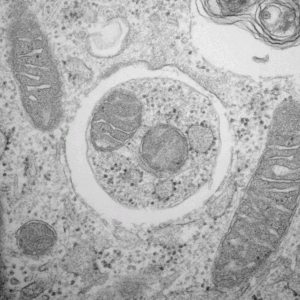
Figure 1: Cytoplasmic components surrounded by an autophagosome,
Adapted by permission from Macmillan Publishers Ltd: Nature Cell Biology 12(9) September 2010, copyright 2010.
Photo taken by Chieko Kishi
In all living organisms, entire cells are replaced over time. Additionally, the cytoplasmic components that make up those cells are also replaced, piece by piece (Figure 1). Autophagy is one of the ways in which this latter piece-by-piece replacement is carried out. At times maintaining the intracellular environment, at times bringing about dynamic change, autophagy is a key process underlying the phenomenon of life and a hot topic in biomedical research. In this process, a type of organelle called an autophagosome surrounds a section of cytoplasm and decomposes it by fusing with a lysosome, another organelle containing digestive enzymes. Pioneered by University of Tokyo Graduate School of Medicine Professor Noboru Mizushima and Tokyo Institute of Technology Frontier Research Center Professor Yoshinori Ohsumi, autophagy is an active field of research throughout the world today.
The great leap forward: yeast autophagy research
At a time when most biomedical researchers were focused on materials synthesis or how things are made, then-Dr. Mizushima’s interest was in metabolism and the cycle from synthesis to degradation and back to synthesis again. The study of intracellular turnover was not yet a well-established discipline, and despite its importance, there were limited research methods available.

Figure 2: Professor Ohsumi’s paper that started the field of autophagy research.
© 2014 University of Tokyo.
The turning point came in 1997 when Dr. Mizushima read a paper by Professor Ohsumi, then professor at the National Institute for Basic Biology. This rather plain-looking paper reported the identification of genes related to autophagy in yeasts, but was the pioneering paper that brought about the emergence of autophagy as an academic field and paved the way for molecular biological approaches when microscope observation was predominant. Convinced that autophagy research would become an important field of research, Dr. Mizushima immediately decided to study under Professor Ohsumi (Figure 2).
After several years of research in Professor Ohsumi’s lab, Dr. Mizushima and his colleagues revealed the function of autophagy-related genes in yeasts. Their research also showed that most of these genes are also conserved in mammals and plants, leading to significant breakthroughs in autophagy research in the years since.
Mammalian autophagy and clinical prospects
As a physician Professor Mizushima recognized from the start that his research could be applied to mammals, but there were no established research methods. This problem was finally overcome by Professor Mizushima’s group in 2004 through the creation of two mouse models: one that displayed fluorescent autophagosomes and one with the autophagy genes knocked out (Figure 3). Using jellyfish genes to produce fluorescence in autophagosomes allowed the real-time observation of living organisms, and the combination of this technique with knockout mice allowed autophagy functionality to be closely investigated.
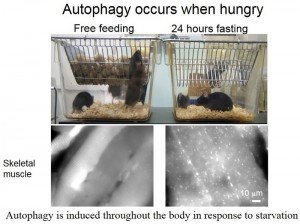
Figure 3: The glowing-autophagosome mouse model created by Professor Mizushima and provided to researchers around the world.
© 2014 Noboru Mizushima.
Mizushima et al. Mol. Biol. Cell (2004).
Studies in yeast had demonstrated “autophagy for change,” the process of recycling existing materials to maintain life or to form spores during starvation, a function that was certainly important in mammal development as well. However Professor Mizushima also discovered another process, that of “autophagy for freshness.” This type of autophagy cleans up rubbish in cells, and if not properly carried out in nerve cells, may lead to the development of neurodegenerative diseases. On the other hand, this means that it should be possible to treat some diseases with drugs that activate autophagy.
Autophagy has also attracted attention as a target for cancer therapy. The idea is to kill tumors by inhibiting the autophagy of cancer cells. This will not be easy because cancer is a disease that has proved resistant to many treatments, but Professor Mizushima emphasizes that if a cancer is found that depends on autophagy more than other cells, it should be possible to develop a magic bullet for that type of cancer.
Joint research and future prospects
It is clear that autophagy research will develop closer links with medicine, but further basic research is also still needed. There remain many research questions to solve at the frontiers of autophagy research, such as the mechanisms and biological significance of selective autophagy, which degrades particular cellular components, and the mechanisms of autophagosome formation.
In part this is why Professor Mizushima moved his laboratory to the University of Tokyo. While electronic means of communication can now link research teams around the world, many studies still originate from face-to-face interaction with connected research teams. Professor Mizushima aims to develop multifaceted approaches in cooperation not only with other medical researchers but also with the graduate schools of science, pharmaceutical sciences, agriculture and life sciences, and engineering.
Even Professor Mizushima, who had anticipated the clinical application of his research from the beginning, humbly declines any responsibility for the tremendous potential that autophagy research has shown, suggesting that the great findings were not due him, but to the importance of autophagy itself. Yet it could be said that Professor Mizushima himself is like the autophagy he studies, switching to Professor Ohsumi’s laboratory and then to the University of Tokyo in search of change. It is certain that there are another ten years of great changes ahead for autophagy research.
Researcher

Professor Noboru Mizushima






