Identification of substrate-capturing chopsticks in γ-secretase Towards development of anti-Alzheimer's treatment without adverse effects

Dementia is one of the biggest social problems in an aging society. Alzheimer’s disease is the most common form of dementia and is thought to be caused by senile plaques created by the massive deposition of aggregation-prone amyloid-β peptide. However, simple inhibition of γ-secretase, which is a responsible enzyme for Aβ production, has been shown to cause adverse effects because γ-secretase is also involved in cleaving other proteins. In addition, γ-secretase is an atypical intramembrane-cleaving protease and its substrate recognition mechanism still remains unknown.
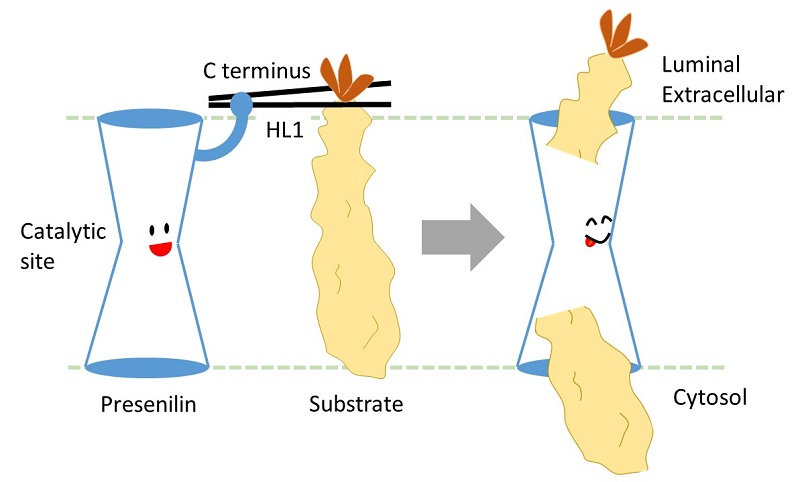
Researchers lead by Professor Taisuke Tomita, and former graduate students Shizuka Takagi-Niidome and Tomoki Sasaki, at the Graduate School of Pharmaceutical Sciences, the University of Tokyo, revealed the mechanism by which presenilin, a catalytic subunit of γ-secretase, captures its substrate in the intramembrane region. They identified that hydrophilic loop 1 and the carboxy terminus of presenilin are cooperatively involved in capturing the extracellular region of the substrate in a similar fashion to chopsticks.
This is the first report to identify the molecular domains involved in substrate capture during γ-secretase-mediated proteolysis (protein cleavage). These findings could lead to the development of anti-Alzheimer treatments that selectively inhibit Aβ production without adverse side effects.
Paper
, "Cooperative roles of hydrophilic loop 1 and the C terminus of presenilin 1 in the substrate-gating mechanism of γ-secretase", The Journal of Neuroscience Online Edition: 2015/2/11 (Japan time), doi: 10.1523/JNEUROSCI.3164-14.2015.
Article link (Publication)
Links
Graduate School of Pharmaceutical Sciences
Department of Neuropathology and Neuroscience, Graduate School of Pharmaceutical Sciences