Molecular mechanism that regulates production of cellular protein degradation machinery Paving the way for development of new cancer drugs

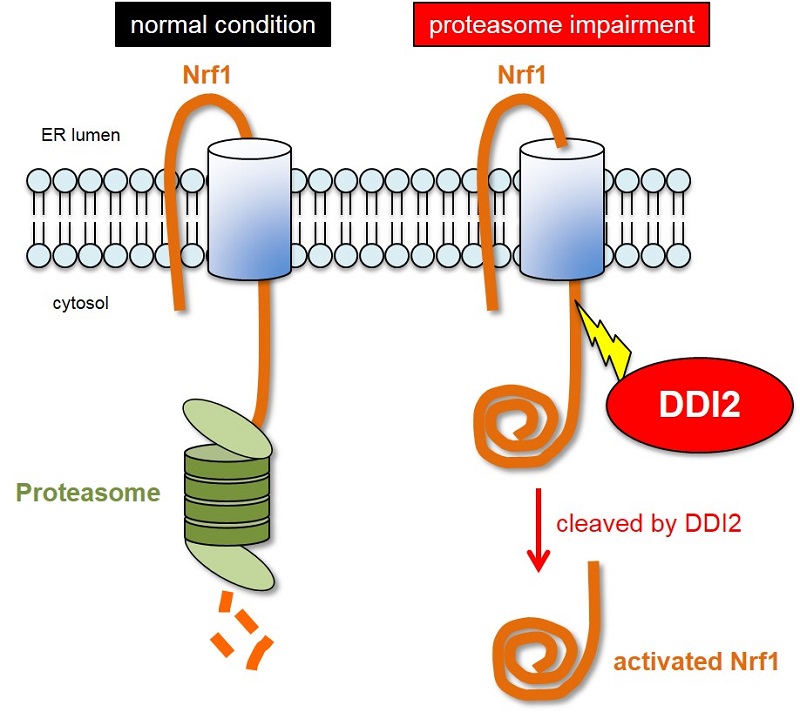
DDI2 promotes production of the proteasome by severing (cleaving) transcription factor Nrf1
In normal conditions, the Nrf1 transcription factor is broken down by the proteasome. However, when the proteasome is impaired, Nrf1 accumulates on the endoplasmic reticulum, causing it to become split off by DDI2. The cleaved, or severed, Nrf1 translocates to the nucleus where it acts as a transcription factor and promotes proteasome gene expression.
© 2016 Shigeo Murata.
Researchers at the University of Tokyo found that an enzyme called DDI2, which breaks down proteins, is necessary for promoting the production of proteasomes—huge protein degradation enzymes composed of multiple proteins. This finding holds promise for the development of new drugs to treat cancer.
The proteasome is a huge molecule within the cell composed of multiple proteins bound to one another, which form the huge protein complex that acts as a gigantic enzyme. It selectively breaks down unneeded or damaged proteins inside the cell, and is necessary for the expression and maintenance of cellular function. Thus, mechanisms for regulating the amount of the proteasome to appropriate levels are essential for maintaining a healthy body. When the proteasome’s function to break down proteins becomes impaired, a transcription factor—a protein that regulates transcription of genetic information—called Nrf1 is activated, increasing the production of the proteasome. However, the precise molecular mechanism for Nrf1 activation remained elusive.
The research group led by Professor Shigeo Murata at the Graduate School of Pharmaceutical Sciences searched for, through comprehensive analysis, the factor responsible for activating the Nrf1 transcription factor in human cultured cells, and succeeded in being the first to reveal that the DDI2 enzyme is needed to activate Nrf1 and promote proteasome production.
While Nrf1-induced production of the proteasome is necessary for breaking down proteins, scientists believe increased proteasome activity contributes to proliferation of cancer cells. In fact, inhibiting proteasome function was shown to be effective in treating multiple myeloma and other cancers.
“Our finding is significant in that it demonstrates a new molecular mechanism for the production of the proteasome,” says Murata. He continues, “It should provide a clue for establishing a new cancer therapy through the development of drugs targeting proteasome production.”
Paper
, "The aspartyl protease DDI2 activates Nrf1 to compensate for proteasome dysfunction", eLife Online Edition: 2016/08/16 (Japan time), doi: 10.7554/eLife.18357.
Article link (Publication)
Links
Graduate School of Pharmaceutical Sciences
Laboratory of Protein Metabolism, Graduate School of Pharmaceutical Sciences






