Beyond antibiotics

This is a series of articles highlighting some of the research projects at the University of Tokyo registered under its Future Society Initiative (FSI), a framework that brings together ongoing research projects that contribute to the United Nations Sustainable Development Goals (SDGs).
FSI Project 036
Research in recent years has shown that gut bacteria are linked not only to digestive system disorders such as ulcerative colitis but to nearly every other condition, including allergies, autoimmune disorders, cancer, diabetes, depression and Alzheimer’s disease. Certain gut bacteria can cause these illnesses by overly stimulating and upsetting our bodies. To prevent this, substances in the gut immune system work to combat and neutralize the harmful bacteria.
As Professor Reiko Shinkura describes it, “The immune system distinguishes foreign invading substances, the nonself, from substances that already exist in our bodies, the self, and then quickly disposes of the invaders. That is what the textbooks tell us. In fact, the immune system also has the ability to not attack benign substances, even if they are recognized as foreign. By researching the mechanisms through which the immune system differentiates target substances to attack, we are aiming for new antimicrobial strategies to antibiotics.”
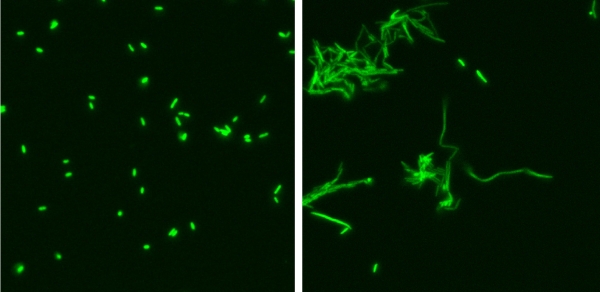
E. coli bacteria are tagged with GFP (green fluorescent protein) to make them visible. In the presence of a certain IgA antibody, E. coli cannot proliferate (Photo 1), whereas in the absence of the antibody, E. coli proliferate rapidly and clump together in “strings” (Photo 2) after three hours of cultivation.
Antibiotics have saved the human race from infectious bacterial diseases such as tuberculosis, and they have been called the greatest discovery of the 20th century. However, they affect nearly all the bacteria we carry within us and bear the risk of causing drastic changes to our body systems. If we had an agent that only attacks harmful bacteria, we might be able to avert that risk.
“IgA antibodies secreted at our mucous membranes protect them from foreign invading substances, whereas if IgE antibodies are produced there, they cause allergic reactions like hay fever. At present, we only have allergy treatments that suppress the functions of IgE antibodies. If we had a treatment that could produce IgA instead of IgE against allergens, we would be able to eliminate the cause of allergies on a fundamental level,” explained Shinkura, whose research has taken initial steps toward creating such a novel treatment.
SDGs supported by this project

Professor Reiko Shinkura │ Institute for Quantitative Biosciences
Related links
- UTokyo FSI

- Health and longevity through improvement of gut microbial condition by intestinal IgA (FSI project page)

- Future Society Initiative Magazine






