Why do people become senile with age?

An introduction to academia starting with “Why?”
After making a list of questions which would pique anyone’s interest, we asked faculty members on campus who might be knowledgeable in these areas to answer them from the perspective of their respective expertise. Let’s take a look into the world of research through questions that you feel you know something about, but cannot answer definitively when actually asked.
Q9. Why do people become senile with age?
Symptoms such as inability to recall people’s names and saying the same thing repeatedly are part of the condition known as dementia. What exactly is going on in the brain of a dementia sufferer? Answered by Taisuke Tomita
Answered by Taisuke TomitaProfessor, Graduate School of Pharmaceutical Sciences
| Functional Pathology |
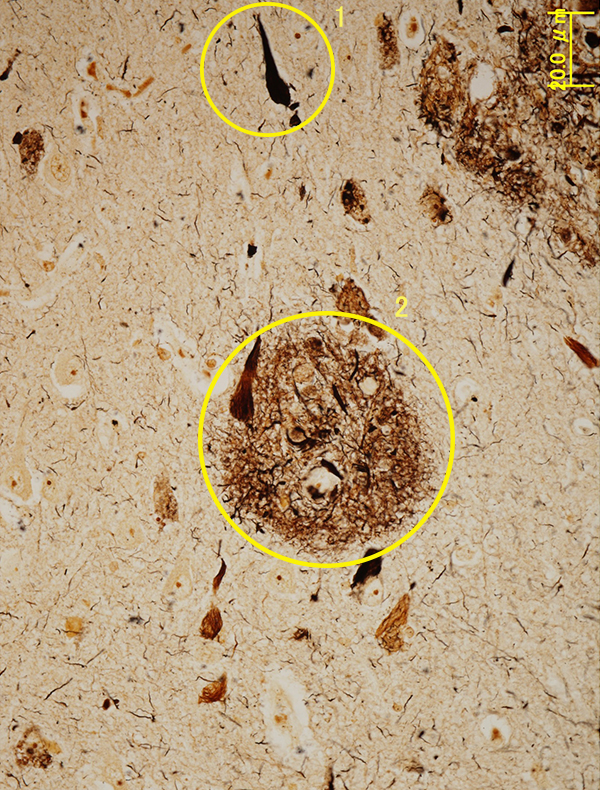
Neurofibrillary tangles are formed by a protein called tau. The accumulation of these tangles in the brain is thought to increase the probability of neural cell death.
Senile plaques
The probability of contracting Alzheimer’s disease in the future is thought to increase with the accumulation in the brain of senile plaques, which are formed by the amyloid beta protein. In order to measure the volume of amyloid beta accumulated in the brain, it was previously necessary to perform highly complicated tests such as brain imaging and cerebral fluid analysis, but advances in testing technology have now made measurement possible using blood samples, and work is progressing toward the practical application of this technique.
Proteins build up in the brain
The major problem in dementia is the decline in cognitive functions such as memory, reasoning and judgment. Communication among neural cells in the brain is what enables us to perform tasks such as remembering things and gives rise to our emotions, but if you examine the brains of people suffering from Alzheimer’s disease, which accounts for the majority of dementia cases, you will find that many of the neural cells have died. For more than a century, researchers have been seeking to understand the mechanisms behind this phenomenon.
Advances in protein analysis technologies, genetic analysis and the like have revealed that neural cell death is caused by accumulation in the brain of senile plaques and neurofibrillary tangles. These two forms of brain “debris” both comprise proteins – amyloid beta in the case of senile plaques and tau in the case of neurofibrillary tangles – which exist in the brain from the time it is first formed. All proteins pass through a repeating cycle of production and destruction, but the metabolism of amyloid beta in particular slows down as you get older, so it builds up more easily in the brain. Our current understanding is that when the accumulation of amyloid beta continues over a long period of time, it results in some kind of stress that causes the tau protein to build up as well. This is what leads to neural cell death.
Using light exposure to break down brain debris
At present there is no method for treating the root cause of Alzheimer’s disease. The difficult thing about diseases of the brain is that once neural cells have died, there is almost no way of reviving them. In recent years a number of research institutions have been looking into ways of clearing the brain of accumulated proteins as a means of treating such diseases before symptoms emerge. My research lab is collaborating with the lab of Professor Motomu Kanai on developing a light-based therapy for dementia. This involves administering a drug that facilitates the breakdown of amyloid beta under light (a photo-oxygenation catalyst), then exposing the brain to light. Side effects are a problem with many drug therapies, but in this case the drug is activated only through light exposure, so it does not cause any harm as it is carried around the body. We hope to begin clinical trials of this therapy within the next few years.

What people can do in everyday life to reduce their risk of cognitive decline is to use their brain while exercising and maintain healthy eating habits. This advice might seem mundane, but we know for sure that it has some effect in lowering the risk. There has also been a focus recently on the relationship with sleep, and experiments on rodents have revealed that debris is cleared from the brain during sleep.
My focus at the moment is on ascertaining why the accumulation of amyloid beta also causes the tau protein to build up. The overall process from the accumulation of amyloid beta to the accumulation of tau to the death of neural cells takes around 10 to 20 years in total, but we still don’t know exactly what is occurring at each stage of this process. And some people don’t experience neural cell death despite having a buildup of amyloid beta and tau. I want to understand why this is so. I believe that if we can uncover the changes that are going on in the brain and body, it will aid both the diagnosis and treatment of Alzheimer’s disease.

Jikken Igaku (“Experimental Medicine”) Vol. 37 (Yodosha, 2019)
Professor Tomita oversaw a special feature in this book titled “The Next Breakthrough in Neurodegenerative Disorders: Brain Environment Homeostasis and Propagation/Elimination of Abnormal Proteins.”
* This article was originally printed in Tansei 45 (Japanese language only). All information in this article is as of September 2022.






