Shedding light on medical therapy for an intractable liver disease Success in drug therapy for progressive familial intrahepatic cholestasis

Progressive familial intrahepatic cholestasis type 2 (PFIC2), an inherited autosomal recessive liver disease caused by mutations in the Bile Salt Export Pump (BSEP) gene, progresses to severe cholestasis (a condition where bile cannot flow from the liver to the intestine), leading to liver failure and death before adulthood. The only therapeutic approach that is currently available is liver transplantation and therefore the development of a new, less invasive and less expensive medical therapy for this disease is of the highest priority.p>
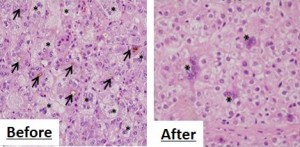
© Hisamitsu Hayashi. Histological analysis (a method of observing morphological changes under the microscope in specimens from living tissue) of liver biopsy specimens of PFIC2 patients before (left) and after (right) clinical study. 4PB therapy markedly relieved bile plug (arrow) and giant cell transformation (asterisk).
A research team including Assistant Professor Hisamitsu Hayashi and Professor Hiroyuki Kusuhara in the Graduate School of Pharmaceutical Sciences at the University of Tokyo, in collaboration with groups in Dokkyo Medical University Koshigaya Hospital and Saiseikai Yokohamashi Tobu Hospital, designed a clinical study (research carried out through trials on human patients for the purpose of disease diagnosis and prevention, improving therapy, or establishing the cause of disease) to investigate the efficacy of 4-phenylbutyrate (4PB), an approved drug for urea cycle disorders, in the treatment of PFIC2 and demonstrated that the therapy with 4PB markedly improved liver function tests without any side effects. Bile plug and giant cell transformation, both of which are histological characteristics of patients with PFIC2, were also relieved with this therapy.
Therefore, after obtaining extended approval for use of 4PB, this therapy would be the preferred choice, instead of liver transplantation, for treating PFIC2 patients, which make it possible to improve quality of life of patients and their family by reducing risk and medical expenses for the treatment.
Press release (Japanese)
Paper
Sotaro Naoi, Hisamitsu Hayashi*, Takeshi Inoue, Ken Tanikawa, Koji Igarashi, Hironori Nagasaka, Masayoshi Kage, Hajime Takikawa, Yuichi Sugiyama, Ayano Inui, Toshiro Nagai, and Hiroyuki Kusuhara,
“Improved liver dysfunction and relieved intractable pruritus after 4-phenylbutyrate therapy in a patient with progressive familial intrahepatic cholestasis type 2”,
The Journal of Pediatrics Online Edition: 2014/2/5, doi: 10.1016/j.jpeds.2013.12.032.
Article link [PDF]
Links
Graduate School of Pharmaceutical Sciences
Department of Pharmacy, Graduate School of Pharmaceutical Sciences






